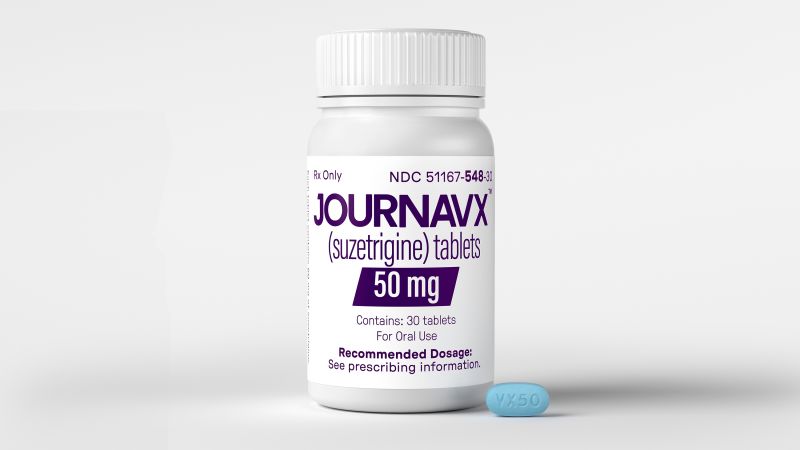
On Thursday, the U.S. Food and Drug Administration (FDA) made a historic decision by approving the first new class of pain reliever in over two decades. This groundbreaking medication is called suzetrigine, which comes in the form of a 50-milligram prescription pill that patients take every 12 hours after an initial larger starter dose. Suzetrigine will be marketed under the brand name Journavx.
Dr. Jacqueline Corrigan-Curay, the acting director of the FDA’s Center for Drug Evaluation and Research, issued a statement highlighting the importance of this non-opioid analgesic. She noted that a new therapeutic class of pain medications for acute pain presents an opportunity to address some of the challenges and risks associated with opioid use. This significant approval reflects the FDA’s dedication to providing patients with safer and effective alternatives for pain management.
Concerning the usage of analgesics, government surveys indicate that these medications are the most frequently prescribed drugs in hospitals across the nation. A staggering statistic reveals that approximately 80 million Americans fill prescriptions annually to manage instances of moderate to severe pain. Notably, about 50% of these prescriptions consist of opioid medications, which carry the inherent risks of dependence and addiction.
Suzetrigine marks a notable return to the development of painkillers, as it is the first new pain medication approved in the U.S. since Celebrex, a type of nonsteroidal anti-inflammatory drug (NSAID), was approved back in 1998. According to Dr. Sergio Bergese, an anesthesiologist at Stony Brook University’s Renaissance School of Medicine, pain sensation involves multiple bodily systems working together. The process starts with nerve cells transmitting electrical signals from the damaged tissue to the brain, which interprets these signals as pain.
In contrast to opioids, which inhibit pain perception in the brain, suzetrigine operates by blocking the initial pain-signaling nerves throughout the body. Dr. Bergese explains that suzetrigine disrupts the pathway of pain signals. Even with existing tissue damage, the brain remains unaware of the pain.
Importantly, suzetrigine does not induce feelings of euphoria or a “high,” features that are often associated with opioid medications. As such, physicians are optimistic that suzetrigine carries a significantly lower risk of addiction or dependence among its users. The drug’s discovery is intriguing; researchers were inspired by a family of fire walkers in Pakistan who demonstrated an unusual inability to sense pain due to a genetic mutation. This unique observation laid the groundwork for developing suzetrigine, culminating in a process that took 25 years to bring to fruition.
Dr. Stephen Waxman, director of the Center for Neuroscience and Regeneration Research at Yale School of Medicine, elaborates on the science behind the medication. Suzetrigine works by closing a specific sodium channel responsible solely for conducting pain signals. The significance of this breakthrough is profound; it not only validates the potential of sodium-channel blockers in pain relief but also paves the way for the development of a second generation of more effective pain management medications.
Clinical trials for suzetrigine have involved nearly 600 participants. In these studies, individuals reported that suzetrigine effectively controlled pain post-abdominal and foot surgeries. The efficacy demonstrated was comparable to that of Vicodin, a combination medication of acetaminophen and hydrocodone. However, the trials were not explicitly designed to pit suzetrigine against Vicodin, complicating conclusions on comparative efficacy.
Results indicated a significant reduction in participants’ pain levels, with an average decrease of about 3.5 points on a scale from 0 to 10, though the medication does not eliminate pain entirely. In a separate study on individuals with sciatica-related back pain, the results suggest that suzetrigine may not excel in treating chronic pain conditions.
Vertex Pharmaceuticals, the company behind suzetrigine, contests these findings, asserting that the drug has demonstrated efficacy in various chronic pain conditions, including diabetic neuropathy—where high blood sugar damages nerves leading to discomfort and more profound complications. Even so, the sciatica trial included a smaller cohort, which raises questions about the statistical power to discern differences between the medication and placebo.
The medical community views suzetrigine’s approval as a valuable addition to the array of pain management options. Experts have expressed their enthusiasm about the new possibilities in treating pain more effectively, citing the need for diverse approaches tailored to individual patients’ needs.
Vertex intends to set the wholesale price at $15.50 per 50-mg pill, with patient assistance programs in place. Nevertheless, reimbursement policies from insurance companies remain unknown, prompting concerns about accessibility for patients who may benefit from this novel medication. Thus, while the scientific community and healthcare providers celebrate this advancement, the future will determine how well this medication integrates into the landscape of pain management.