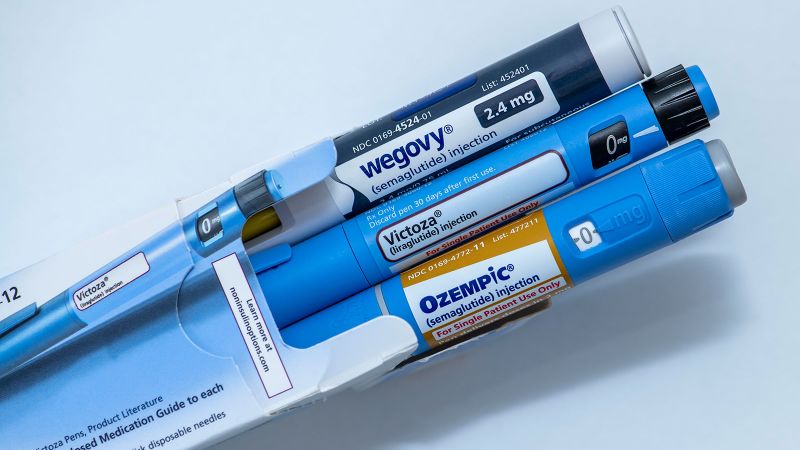
On Monday, the United States Food and Drug Administration (FDA) announced its approval of a generic form of liraglutide, a daily injectable medication commonly used for managing type 2 diabetes. This decision not only introduces a more affordable alternative to patients but also aims to alleviate an ongoing shortage of the drug. The approval is particularly significant as it opens avenues for lower-cost options that can help a large number of individuals managing diabetes.
Liraglutide, often marketed under the brand name Victoza, was among earlier entries in the glucagon-like peptide-1 (GLP-1) medication class, which includes the more recently developed semaglutide, known for its brand Ozempic. Both these medications are produced by Novo Nordisk, a prominent pharmaceutical company based in Denmark. The introduction of generic liraglutide by Hikma Pharmaceuticals USA presents a welcome relief, as the company has indicated that availability across the nation is anticipated before the year’s end.
The pricing details for the generic liraglutide remain undisclosed, but representatives from Hikma have ensured that it would be less expensive compared to the branded Victoza. According to Novo Nordisk, the cost for branded liraglutide can range from $500 to $815 per package, varying depending on the prescribed dosage. It’s important to note that Novo Nordisk also offers another brand of liraglutide designed for treating obesity, known as Saxenda.
Healthcare professionals have noted the importance of this generic approval in light of the increasing number of individuals relying on medications like liraglutide. Dr. Harlan Krumholz, a cardiologist affiliated with Yale University, stated that the approval of a second generic version is likely to benefit many patients. However, he highlighted a potential concern regarding the efficacy of treatment options available to patients, citing that newer GLP-1 medications, which allow for weekly injections rather than daily, have demonstrated greater effectiveness for individuals struggling with obesity and other related conditions. Unfortunately, these newer options are not yet available in generic form.
In addition to liraglutide, the market includes other GLP-1 drugs such as Ozempic and Wegovy, which utilize semaglutide, along with Mounjaro and Zepbound, which utilize tirzepatide. These medications can be notably expensive, often exceeding $1,000 per month without insurance or discounts, prompting concerns about a two-tiered healthcare system. Dr. Krumholz warned that less affluent patients may find themselves limited to options that lack robust evidence of benefits.
The introduction of an authorized generic from Teva Pharmaceuticals in June further complicates the landscape. This version, which was created under a settlement agreement with Novo Nordisk, is essentially the same as the branded product but sold without the brand name, being slightly cheaper—approximately 14% lower than branded Victoza.
The FDA differentiates between traditional generic drugs, which are developed, manufactured, and marketed by another company, and authorized generics. Distinct from these, compounding pharmacies can produce personalized drug formulations and have proliferated in response to shortages. However, compounded medications are less rigorously regulated, prompting safety and efficacy concerns.
The FDA’s approval of generic liraglutide is particularly noteworthy as the drug has been in shortage in the U.S. since July 2023. With its recent announcement, the FDA emphasized prioritizing generic drug applications for medications experiencing shortages, ensuring that patients have better access to essential therapies.
Healthcare professionals are hopeful that the price of generic liraglutide will reflect significant savings compared to its branded counterparts. Dr. Jody Dushay, an endocrinologist at Beth Israel Deaconess Medical Center, expressed her optimism about the upcoming availability of generic liraglutide. However, she acknowledged that the dynamics of the pharmaceutical market often indicate that pricing reductions can take time and may not yield immediate results.
The development suggests a positive shift toward improved accessibility for patients requiring diabetes treatment. Yet, the real achievement will depend on the efforts of multiple manufacturers entering the market, as competition among generics can dramatically lower prices and expand treatment options for patients in need.